Navigating the 'Orphan Trap'


The pharmaceutical landscape in March 2026 has reached a point of divergence where the biological capability to engineer therapies for the rarest of human conditions has surpassed the traditional economic frameworks designed to support drug commercialization. This phenomenon, increasingly characterized as the "Orphan Trap," describes a systemic failure where small biotechnology firms successfully navigate the complexities of molecular discovery only to be ensnared by the unsustainable costs of a commercial infrastructure designed for mass-market or even broad orphan indications. For therapies targeting ultra-rare populations defined as those affecting significantly fewer than the 200,000-patient threshold established by the Orphan Drug Act of 1983 the traditional commercial playbook is not merely inefficient; it is functionally obsolete.
As of early 2026, progress in data science and an increased understanding of disease genetics have led to the identification of more than 10,000 rare diseases affecting over 30 million people in the United States alone. Despite this aggregate scale, the individual patient populations for many newly druggable genetic defects are so small that traditional randomized controlled trials (RCTs) and conventional sales forces are no longer viable. Navigating this environment requires a radical redesign of the biotech commercial team, shifting away from volume-based reach toward high-precision, science-led engagement models that leverage the latest regulatory flexibilities and real-world evidence frameworks provided by the U.S. Food and Drug Administration (FDA).
The Regulatory Paradigm Shift: The Plausible Mechanism Framework
A primary pillar in overcoming the "Orphan Trap" is the utilization of newly established regulatory pathways that lower the evidentiary burden for small biotechs while maintaining rigorous safety standards. On February 23, 2026, the FDA issued a landmark draft guidance introducing the "Plausible Mechanism Framework". This framework represents a transformative shift for individualized therapies, particularly genome editing (GE) and RNA-based therapies such as antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs).
The "Plausible Mechanism Framework" allows sponsors to generate substantial evidence of effectiveness and safety when randomized clinical trials are not feasible due to extremely small patient populations. By focusing on therapies for diseases caused by identifiable genetic variants with a clear mechanistic rationale, the agency permits sponsors to rely on the underlying cellular or molecular biology of the disease. This is often described as a "GPS" approach: once the platform (the delivery vector or backbone) is established, the specific genetic "address" (the sequence) can be modified for individual patients without requiring a ground-up clinical trial for every new variant.
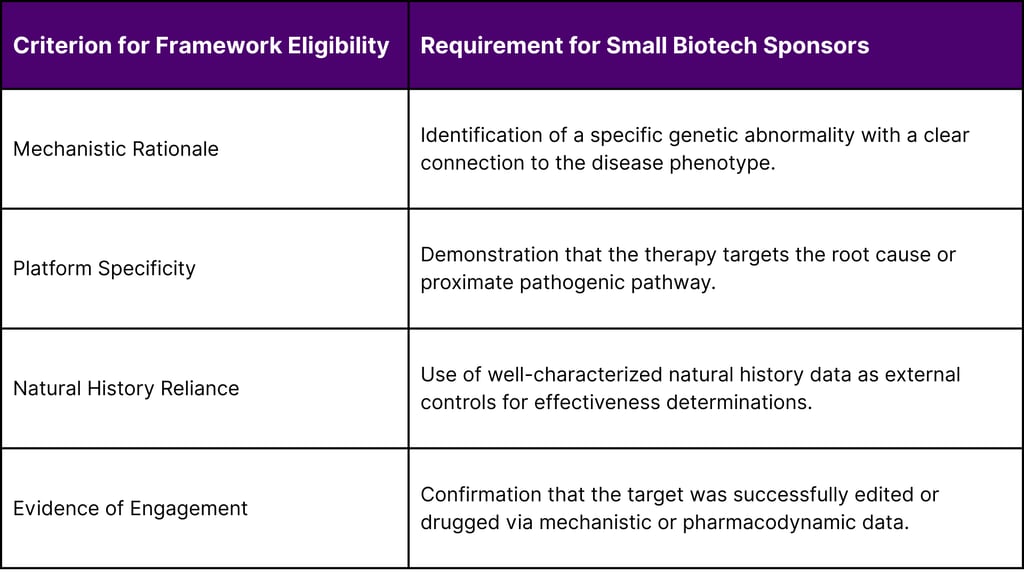
The implications for a lean commercial team are significant. Historically, a substantial portion of a biotech’s early commercial investment was diverted into clinical development support and patient identification for large trials. Under the Plausible Mechanism Framework, the burden shifts toward natural history data collection and the establishment of robust, de-identified databases. The agency has indicated a new willingness to accept real-world evidence (RWE) from sources such as national cancer registries, hospital databases, and insurance claims without requiring identifiable individual patient data in all cases. This allows small teams to focus their resources on data stewardship and partnerships with academic medical centers rather than the logistics of multi-site RCTs.
The Rare Disease Innovation Hub and the 2026 Strategic Agenda
In tandem with these new frameworks, the FDA’s Rare Disease Innovation Hub (the Hub) has emerged as a centralized gateway for small biotechs. Launched to coordinate efforts between the Center for Biologics Evaluation and Research (CBER) and the Center for Drug Evaluation and Research (CDER), the Hub’s 2026 Strategic Agenda, released on February 2, 2026, outlines clear goals for external partner engagement.
The Hub received dedicated funding for 2026 $250,000 from CBER and $750,000 from CDER to support activities intended to streamline the development pathway.9 For a small biotech with limited bandwidth, the Hub serves as a "First Stop" for navigating various regulatory science initiatives. Key initiatives under the 2026 Strategic Agenda include:
Rare Disease Roadmap: A web-based navigation tool designed to help developers identify the appropriate programs and points of contact across the agency.
RISE Workshop Series: The "Rare disease Innovation, Science, and Exploration" workshops, which focus on issues like control arm options for small populations and the promises of AI in drug discovery.
Quarterly Newsletters and Consolidation: Efforts to streamline rare disease program information onto a single, accessible platform, reducing the research burden on lean commercial teams.
Furthermore, the "Rare Disease Evidence Principles" (RDEP) initiative, introduced in 2025, provides a structured mechanism for biotechs to seek assurance that the agency will consider additional supportive data for ultra-rare conditions. Programs meeting RDEP eligibility can secure an additional meeting with review teams early in development, allowing lean teams to align scientific strategy with commercial expectations before substantial capital is deployed.
Analysis of the 2025 Market Landscape and 2026 Projections
The market context for 2026 is defined by the high success rate of orphan-designated therapies in the previous year. In 2025, CDER approved 46 novel drugs, 23 of which (50%) carried an Orphan Drug Designation.12 When combined with CBER approvals, the total number of novel drug approvals reached 58 for the year.
2025 Novel Drug Approvals Statistics
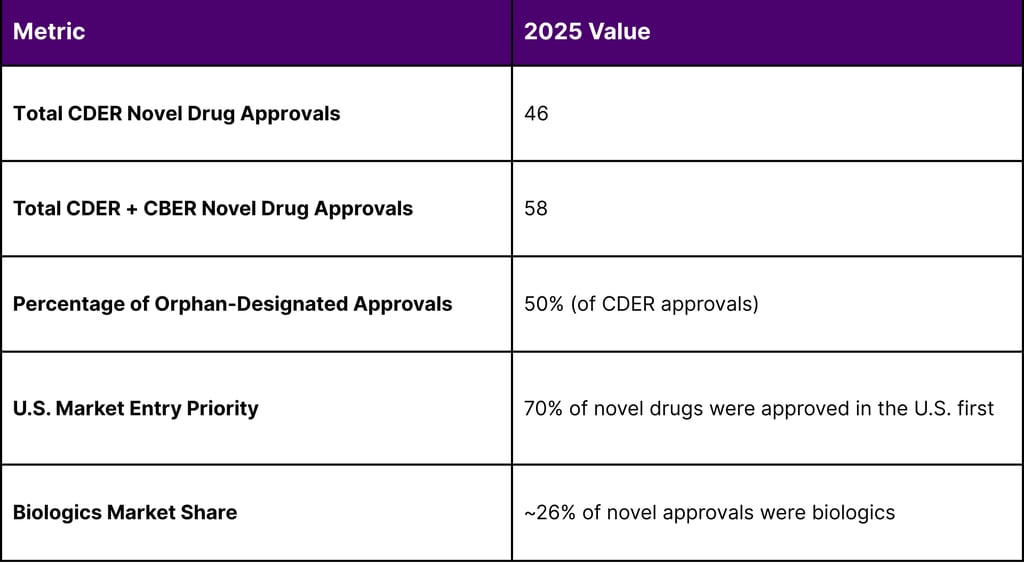
The prevalence of biologics monoclonal antibodies, gene therapies, and recombinant proteins is particularly relevant for ultra-rare indications. In 2025, biologics accounted for 65.8% of the global orphan drugs market share. This dominance is expected to increase, with forecasts suggesting that biologics and gene therapies may account for over 60% of new drug approvals by 2030. For a small biotech, this necessitates a commercial team that is heavily weighted toward medical affairs and complex supply chain management rather than consumer-style marketing.
The global orphan drugs market was valued at USD 261.0 Billion in 2025 and is projected to reach USD 589.2 Billion by 2034, exhibiting a CAGR of 9.47% from 2026-2034. Despite this growth, the "Orphan Trap" remains a risk because much of this revenue is concentrated in larger orphan indications such as oncology (which holds a 36.6% market share) and cystic fibrosis. Ultra-rare genetic disorders often face "market failure" where high development costs intersect with extremely small patient numbers, requiring a pricing sensitivity analysis. For example, a treatment for an ultra-rare disease affecting only 200 people per year that costs $1B to develop would requires a sustainable price of approximately:
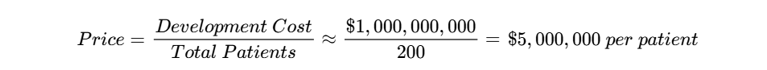
Actual pricing frameworks are sensitive to volume; if the patient population were 10,000, the price could drop significantly to roughly $132,699 per patient. This underscores the critical importance of a lean infrastructure to minimize the overhead component of the price point.
Strategic Advice: Building the Lean Commercial Team
To avoid the "Orphan Trap," small biotechs must adopt a "Rare Mosaic" approach to market intelligence and team construction. This methodology emphasizes segmenting fragmented and opaque markets to identify "high-potential micro-markets" regions or centers of excellence where patients are most likely to be diagnosed and treated.
The Core Roles of a Lean Team
The conventional sales force is replaced in 2026 by a hybrid "Scientific Liaison and Access" model. The lean commercial team should be structured around three primary functions:
Scientific Liaison Officers (SLOs): These roles go beyond the traditional Medical Science Liaison (MSL). SLOs are responsible for "Strategic Audience Mapping," identifying the very small number of physicians (often globally) who manage the target patient population. They focus on scientific education and the molecular mechanism of action, ensuring that clinicians understand the "Plausible Mechanism" established during regulatory review.
Patient Success Navigators: In ultra-rare indications, the patient journey is the primary commercial barrier. Navigators work within a "Launch Sphere RX" framework to evaluate market readiness and product fit for individual patients. They handle the high-touch coordination required between payers, specialized pharmacies, and treatment centers.
Market Access & RWE Specialists: This function manages the "Compliance & Risk Solutions" necessary for global approval pathways. They are responsible for communicating the value proposition to payers, utilizing natural history data and biomarkers to justify premium pricing in the absence of traditional Phase III RCT data.
Leveraging the I-Corps and NCATS Ecosystem
Small biotechs should not build these capabilities in a vacuum. The National Institutes of Health (NIH) provides several "customer discovery" and commercialization support programs that can act as an extension of a lean team.
The "I-Corps at NIH" program is a hands-on entrepreneurship training initiative for companies that have received Small Business Innovation Research (SBIR) or Small Business Technology Transfer (STTR) funding. The 2026 cohort schedule provides specific windows for biotechs to hone their target markets and build the skills required for intensive stakeholder engagement.
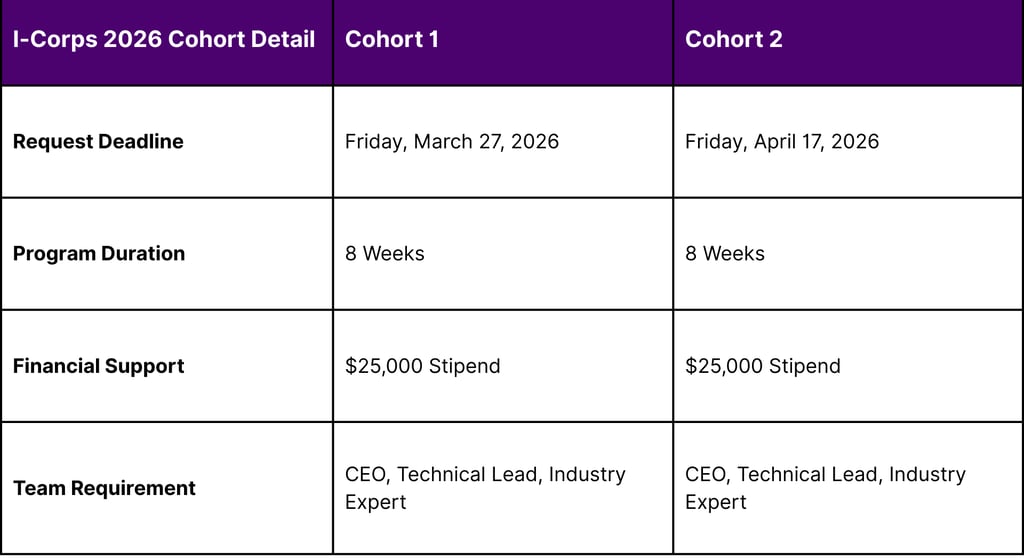
Additionally, the "Commercialization Readiness Pilot (CRP) Program" provides awarded Phase II and Phase IIB small businesses with technical assistance and funding for late-stage development that is not covered by standard grants. By utilizing these federal "seed" funds and technical assistance programs (TABA), a small biotech can effectively outsource a significant portion of its early-stage commercial strategy and market research.
Market Access and the 2026 Reimbursement Landscape
The commercial success of an ultra-rare therapy is ultimately determined by its integration into the Medicare and Medicaid systems. The 2026 Medicare Physician Fee Schedule (PFS) Final Rule, which became effective on January 1, 2026, introduced several changes that impact how "bespoke" therapies are administered and reimbursed in clinical settings.
Medicare Part B Conversion Factors and APMs
CMS has finalized two separate conversion factors for 2026, creating a financial "nudge" toward Alternative Payment Models (APMs).
Qualifying APM Conversion Factor: $33.57 (a projected increase of 3.77% from 2025).
Non-Qualifying APM Conversion Factor: $33.40 (a projected increase of 3.26% from 2025).
For biotechs, this means that the clinics and hospitals most likely to administer complex, individualized therapies such as specialized academic medical centers are likely to be part of APMs that prioritize value-based care. Lean commercial teams must be prepared to integrate their therapies into these models, emphasizing long-term outcomes and reduced downstream medical costs.
The 340B Rebate Model Pilot Program
A significant potential hurdle for ultra-rare drug commercialization in 2026 is the HRSA Office of Pharmacy Affairs’ 340B Rebate Model Pilot Program. The 340B program allows covered entities to purchase outpatient drugs at significant discounts. Historically, this was an upfront discount; however, HRSA is exploring a "rebate model" where manufacturers would pay rebates after the purchase.
Small biotechs must carefully monitor this transition, as a rebate model could significantly impact the cash flow of the specialized safety-net providers that often serve as the primary treatment centers for rare disease patients. HRSA is currently seeking input through a Request for Information (RFI) with a deadline of April 20, 2026. Lean commercial teams should engage in this process to advocate for implementation strategies that do not inadvertently create barriers to patient access for low-volume, high-cost therapies.
Protecting Market Exclusivity: The OBBBA and Negotiation Protections
The 2025 Tax and Budget Reconciliation Law (often called the One Big Beautiful Bill Act or OBBBA) provided a critical reprieve for small biotechs by expanding the orphan drug exclusion from Medicare price negotiation.
Under the original Inflation Reduction Act (IRA), only drugs approved for a single rare disease were excluded from negotiation. The 2025/2026 framework broadens this to include drugs designated for multiple rare diseases, provided all approved indications are for rare conditions.

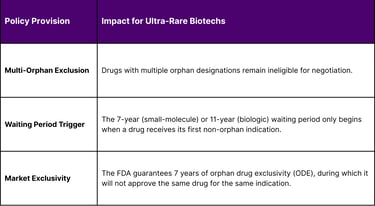
This legislative change is a double-edged sword. While it protects the high prices necessary for ultra-rare biotechs to survive, it also delays the entry of lower-cost generics or biosimilars. For a small biotech, this protection is the "ultimate moat strategy," allowing a lean team to focus on maximizing the clinical value of their first indication while strategically planning subsequent rare-indication expansions without the immediate threat of price caps.
The "Orphan Trap" of Supply Chain and Manufacturing
In 2026, the "Orphan Trap" is not just a commercial or regulatory problem; it is a manufacturing one. The cost of goods (COGS) for bespoke therapies remains high CAR-T cell therapies, for instance, are estimated to cost between $60,000 and $90,000 per dose to manufacture. For an ultra-rare indication with 200 patients, the fixed costs of maintaining a specialized manufacturing facility can be prohibitive.
Small biotechs are increasingly turning to "Platform Vector Gene Therapy" (PaVe-GT) strategies, which are supported by NCATS and the Bespoke Gene Therapy Consortium (BGTC). These initiatives aim to create standardized platforms for AAV gene therapy translation, allowing multiple rare disease therapies to share a common manufacturing backbone. This cross-disciplinary approach is essential for lean teams to prevent their operational costs from spiraling out of control.
Conclusion: Strategic Recommendations for Small Biotechs
Successfully navigating the "Orphan Trap" in 2026 requires a lean commercial infrastructure that is as innovative as the molecular biology it supports. The shift from broad market reach to high-precision engagement is the only viable pathway for ultra-rare indications.
Adopt a "Scientific Liaison" Field Force: Prioritize SLOs and Patient Success Navigators who can identify "high-potential micro-markets" and guide patients through the fragmented diagnosis-to-treatment journey.
Maximize Regulatory Flexibility: Use the Plausible Mechanism Framework and RDEP pathways to reduce the clinical data burden and focus on RWE and natural history data as the primary drivers of effectiveness.
Engage the Innovation Hub Early: Utilize the Hub as a centralized point of contact to align strategies across CBER and CDER, ensuring that regulatory requirements are harmonized before launch.
Leverage Federal Commercialization Support: Participate in I-Corps at NIH and the CRP programs to outsource "customer discovery" and business model validation, preserving capital for critical clinical milestones.
Strategically Manage Exclusivity: Utilize the expanded negotiation exclusions of the OBBBA to protect premium pricing, while avoiding non-orphan indications that would trigger the Medicare price negotiation clock.
By building a commercial infrastructure that is nimble, science-led, and integrated into the federal innovation ecosystem, small biotechs can move beyond the "Orphan Trap" and deliver transformative therapies to the millions of patients currently living with untreated rare genetic disorders.
FAQ
What is the "Orphan Trap"?
It is a scenario where the cost of building traditional commercial infrastructure is too high for the small patient populations found in ultra-rare diseases. Small biotechs must use lean models to remain viable.
How does the Plausible Mechanism Framework help small biotechs?
It allows the FDA to approve individualized therapies (like ASOs) based on a "GPS" approach proving the platform works and focusing on the genetic sequence of the individual patient, rather than requiring new RCTs for every mutation.
What are the 2026 Medicare conversion factors?
For 2026, the APM conversion factor is $33.57 and the non-APM factor is $33.40. This encourages providers to use value-based care models.
Are orphan drugs protected from Medicare price negotiation?
Yes, under the 2025/2026 OBBBA changes, "multi-orphan" drugs that are only designated for rare conditions are excluded from negotiation. The negotiation clock only starts if a non-orphan indication is added.
What is the I-Corps at NIH program?
It is an 8-week training program that helps biotech teams validate their business models and conduct "customer discovery" with a $25,000 stipend to support the process.
References
Rare Diseases at FDA
FDA Proposes New Approval Pathway for Individualized Ultra-Rare Disease Therapies
Orphan Drugs Market Size And Global Trends Report | 2034 - IMARC Group
Genetic and Rare Diseases Information Center | GARD
FDA Rare Disease Innovation Hub
WTAS: FDA Launches Framework for Accelerating Development of ...
FDA Advances a “Plausible Mechanism” Framework for Rare Disease Drug Development and Signals a Shift to a Single Trial with Confirmatory Evidence Default for All Drugs | Advisories | Arnold & Porter
Rare disease round-up: FDA efforts to advance treatments - Hogan Lovells
Rare Disease Month Developments, Part 1 – The Good: RPD PRV ...
Rare Disease Innovation Hub Mission Strategic Agenda 2026 - FDA
Rare Disease Therapies: Four Trends to Watch - Blue Matter Consulting
FDA CDER Releases 15th Annual New Drug Therapy Approvals Report for 2025
Novel Drug Approvals for 2025 | FDA
The Pharmaceutical Industry in 2025: An Analysis of FDA Drug Approvals from the Perspective of Molecules - PMC
The 2026 Biologics Wave: Which Innovations Will Break Through? | Contract Pharma
Orphan Drug Market Consulting Insights 2033 - SkyQuest Technology
A roadmap for affordable genetic medicines - eScholarship
Fyreignis Market Research | Reports & Strategy for Rare Diseases ...
Resources - National Institute of Mental Health (NIMH) - NIH
NIH Entrepreneurship News: Phase I Commercialization Support: I-Corps at NIH Program Webinar, NCI SBIR Informational Webinar, America's Seed Fund Week | 2024 Virtual Road Tour - GovDelivery
Understanding SBIR and STTR | Seed
Calendar Year (CY) 2026 Medicare Physician Fee Schedule Final Rule (CMS-1832-F)
Federal Register :: Medicare and Medicaid Programs; CY 2026 Payment Policies Under the Physician Fee Schedule and Other Changes to Part B Payment and Coverage Policies
Medicare Physician Fee Schedule Final Rule 2026 - ChartSpan
Request for Information: 340B Rebate Model Pilot Program - Regulations.gov
340B Drug Pricing Program - HRSA
340B Rebate Model Pilot Program - HRSA
Request for Information: 340B Rebate Model Pilot Program - Federal Register
CMS Releases Final Guidance for Initial Price Applicability Year 2028
People with Medicare Will Face Higher Costs for Some Orphan ...
The 1825-day head start: How to stop a generic without a patent - DrugPatentWatch
National Center for Advancing Translational Sciences
Rare Disease Day at NIH | National Center for Advancing Translational Sciences
Our Impact on Rare Diseases | National Center for Advancing ...

