The ASO 2.0 Revolution


The landscape of precision medicine has reached a definitive turning point in March 2026, as the maturation of antisense oligonucleotide (ASO) technology converges with a transformative regulatory environment to address the most challenging rare genetic disorders. Once considered a volatile and niche therapeutic modality, ASOs have evolved through three distinct generations of chemical refinement, culminating in what industry experts now term the ASO 2.0 Revolution. This era is characterized by sophisticated sugar and backbone modifications that have effectively resolved historical hurdles such as rapid nuclease degradation, poor cellular internalization, and systemic toxicity. The recent issuance of the U.S. Food and Drug Administration (FDA) Plausible Mechanism Framework in February 2026 serves as the institutional cornerstone of this revolution, providing a streamlined pathway for individualized therapies where traditional randomized trials are mathematically impossible.
The Chemical Architecture of ASO 2.0
The progression from early experimental sequences to the robust pharmacotherapeutic entities of 2026 is rooted in a sequence of chemical innovations. The first generation of ASOs utilized phosphorothioate (PS) linkages, where a non-bridging oxygen atom in the phosphodiester backbone was replaced with a sulfur atom. This modification was pivotal for enhancing resistance to nucleases and improving plasma protein binding, which delayed renal clearance and extended the drug's half-life to approximately 2–3 days. However, the PS backbone alone introduced limitations, including a minor reduction in binding affinity and the potential for pro-inflammatory responses through the activation of Toll-like receptors (TLRs) and the complement system.
The transition to the second generation, the bedrock of ASO 2.0, introduced 2'-ribose sugar modifications such as 2'-O-methyl (2'-OMe) and 2'-O-methoxyethyl (2'-MOE). these modifications imparted superior binding affinity and metabolic stability, significantly widening the therapeutic window. By March 2026, the clinical standard has shifted toward third-generation chemistries, including locked nucleic acids (LNA), constrained ethyl (cEt) modifications, and phosphorodiamidate morpholino oligomers (PMO). LNAs are particularly notable for their "locked" ribose ring, which is bridged by a methylene bond between the 2'-oxygen and 4'-carbon, providing unparalleled thermodynamic stability and nuclease resistance even under extreme physiological conditions.
Comparative Analysis of ASO Chemical Generations and Properties
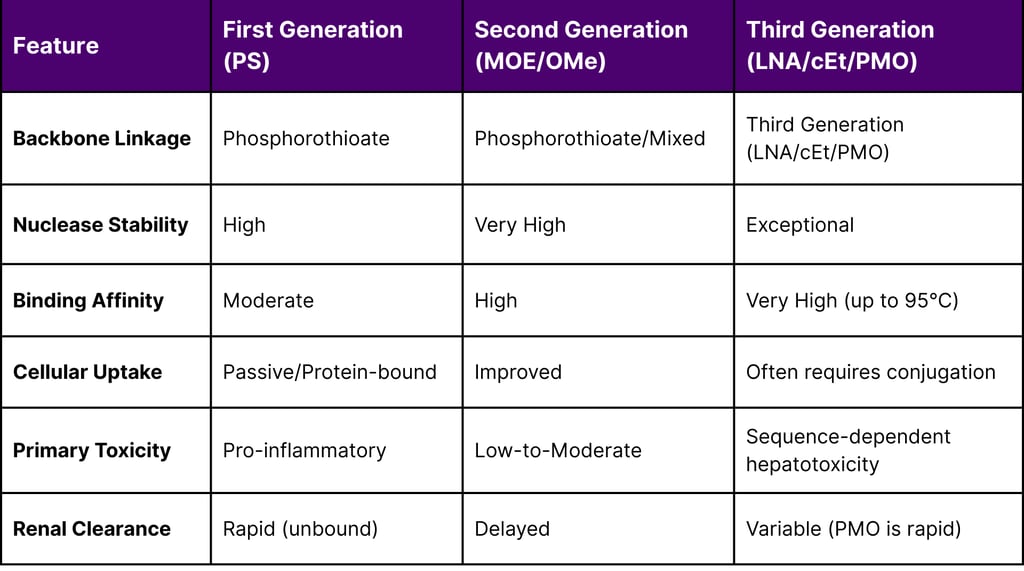
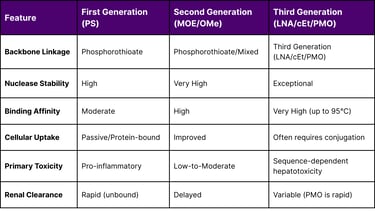
The integration of these chemistries allows for the design of "gapmers," which are chimeric oligonucleotides with a central DNA "gap" that supports RNase H1 activity, flanked by modified "wings" that enhance binding and stability. This architectural flexibility is critical for treating rare diseases like Hereditary Transthyretin-mediated Amyloidosis (hATTR) and SOD1-related Amyotrophic Lateral Sclerosis (ALS), where robust protein knockdown is required.
Mechanistic Diversification in Rare Disease Therapy
The dominance of ASOs in the 2026 rare disease market is further explained by their multi-modal mechanisms of action. Unlike traditional drugs, ASOs can be engineered to either degrade problematic RNA, modulate the splicing of pre-mRNA, or physically block the translation of toxic proteins.
RNase H1-Dependent Pathways
The most common therapeutic approach for gain-of-function mutations involves the recruitment of RNase H1, an endogenous endonuclease that recognizes DNA-RNA heteroduplexes and cleaves the RNA strand. This mechanism is leveraged by several FDA-approved drugs as of March 2026, including Eplontersen (Wainua) and Tofersen (Qalsody). The precision of RNase H1 cleavage depends on a minimum stretch of four DNA nucleotides, with most modern gapmers utilizing 8–10 central nucleotides to maximize catalytic efficiency.
Splice-Switching and Restorative Mechanisms
For diseases caused by loss-of-function or truncated proteins, ASOs act as splice-switching oligonucleotides (SSOs). By binding to specific motifs such as intronic splicing silencers (ISS), ASOs can prevent the exclusion of essential exons or promote the skipping of mutated ones to restore the reading frame. This approach has been revolutionary for Spinal Muscular Atrophy (SMA), where ASOs promote the inclusion of exon 7 in the SMN2 gene, and for Duchenne Muscular Dystrophy (DMD), where exon skipping allows for the production of a truncated but functional dystrophin protein.
The versatility of ASOs also extends to the nucleus, where they can modulate pre-mRNA processing before transcripts are exported to the cytoplasm, a capability that distinguishes them from double-stranded siRNAs. Recent research in March 2026 indicates that covalent conjugation with small-molecule nuclear importers, such as (+)-JQ1, can further improve target engagement in the nucleus, potentially reviving drugs that previously failed in clinical trials due to poor nuclear localization.
Breakthroughs in Delivery Mechanisms
The "Revolution" in the report title refers largely to the maturation of delivery ligands and non-viral vectors that have expanded the reach of ASOs beyond the liver. Historically, systemic administration resulted in the majority of the drug accumulating in the liver and kidneys, limiting the treatment of neurological, pulmonary, and muscular disorders.
Hepatocyte Targeting via GalNAc
The conjugation of N-acetylgalactosamine (GalNAc) to ASOs has become the gold standard for liver-directed therapies. GalNAc serves as a high-affinity ligand for the asialoglycoprotein receptor (ASGPR), which is expressed at high densities (up to receptors per cell) on the surface of hepatocytes. This ligand-mediated endocytosis allows for clinical doses to be reduced significantly often by an order of magnitude thereby minimizing the risk of systemic side effects. As of March 2026, the success of Eplontersen (Wainua) and Olezarsen (Tryngolza) validates this approach, with Wainua demonstrating a robust 45 mg once-monthly subcutaneous dosing regimen.
Extrahepatic and CNS Delivery Systems
For neurological rare diseases, the focus has shifted to overcoming the blood-brain barrier. While intrathecal administration remains the primary route for drugs like Nusinersen and Tofersen, next-generation delivery platforms are emerging. The "Mini-nucleosome" platform, a non-viral vector designed to deliver RNA to the CNS, retina, and lungs, has shown remarkable stability remaining viable at room temperature for over 30 days and at for more than six months. Furthermore, researchers are exploring "free uptake" mechanisms where cEt-modified ASOs enter cells without transfection reagents, aided by endosomal Rab GTPases like Rab5C.
Nanoparticulate and Polymeric Formulations
Advances in polymeric nanoparticles, such as PLGA and PBAE, continue to offer alternatives for localized delivery. While cationic formulations were previously limited by toxicity, newer neutral and biocompatible polymers are being used to create "proton sponge" effects that facilitate endosomal escape, ensuring the ASO reaches the cytoplasm where it can engage with target RNA.
Updated Safety Profiles and Toxicological Refinement
The safety profile of ASOs in 2026 reflects a transition from managing general class effects to a granular understanding of sequence-specific and chemistry-dependent toxicities. The most significant advancement in this area is the use of bioinformatics and machine learning to "screen out" sequences with high off-target potential before clinical entry.
Hybridization-Dependent Off-Target Effects
Off-target binding occurs when an ASO hybridizes with a transcript other than its intended target, often within introns, leading to unintended RNase H cleavage. Research suggests that LNA and cEt gapmers are particularly prone to this if not carefully designed, as their high binding affinity can tolerate mismatches. To mitigate this, modern ASO designs are often shortened to 12–15 nucleotides; shorter sequences have a lower statistical probability of finding near-perfect matches elsewhere in the transcriptome.
Class-Specific Safety Findings in 2024–2026
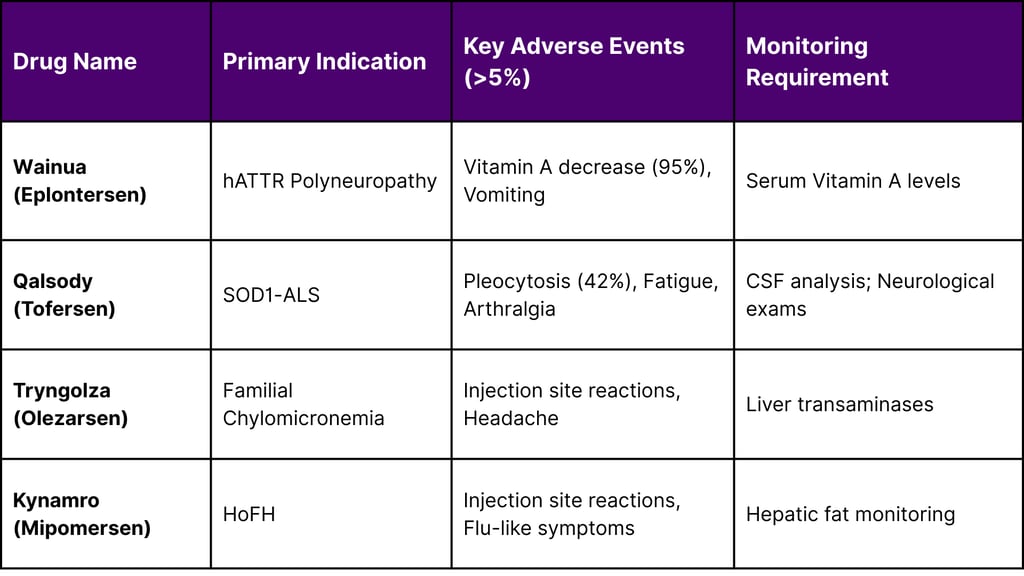
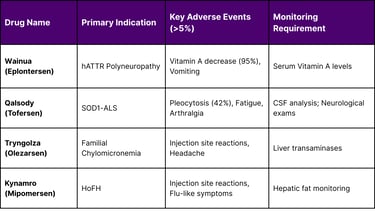
Recent approvals have provided critical data on long-term safety. For example, Eplontersen (Wainua) clinical data (Study 1, N=144) revealed that 95% of patients developed low serum Vitamin A levels because the drug reduces transthyretin, the carrier protein for retinol.9 Consequently, daily Vitamin A supplementation has become a mandatory part of the therapeutic protocol.
In the case of Tofersen (Qalsody) for SOD1-ALS, the Phase III VALOR trial (N=108) identified pleocytosis (increased white blood cell count in the CSF) in 42% of treated patients, along with more severe but rare events such as myelitis and aseptic meningitis. Despite these risks, the FDA granted accelerated approval based on a 33% reduction in total CSF SOD1 protein and significant decreases in neurofilament light chain (NfL), a biomarker of neuronal damage.
Adverse Reactions and Monitoring Statistics (Reported 2025-2026)
Regulatory Evolution: The February 2026 Framework
A pivotal moment in the ASO 2.0 revolution occurred on February 23, 2026, when the FDA launched a new framework for accelerating the development of individualized therapies for ultra-rare diseases. This draft guidance, titled Considerations for the Use of the Plausible Mechanism Framework, acknowledges that for many genetic conditions, the patient population is so small that randomized controlled trials are not feasible.
The Plausible Mechanism Framework
The framework allows sponsors to generate substantial evidence of effectiveness using a combination of nonclinical data, small clinical datasets, and natural history comparisons. Key requirements include:
Identification of Root Cause: The sponsor must identify the specific molecular abnormality (e.g., a unique point mutation).
Drugging the Target: Evidence must confirm that the ASO successfully interacts with the target RNA or restores the functional protein.
Master Protocols: The FDA now encourages the use of master protocols, where a single product application can include variations of an ASO targeting different mutations in the same gene.
This regulatory flexibility is expected to drastically lower the cost and time required to bring N-of-1 therapies to the clinic, making the "Milasen" model of personalized ASOs a scalable reality.
Market Dynamics and March 2026 Status
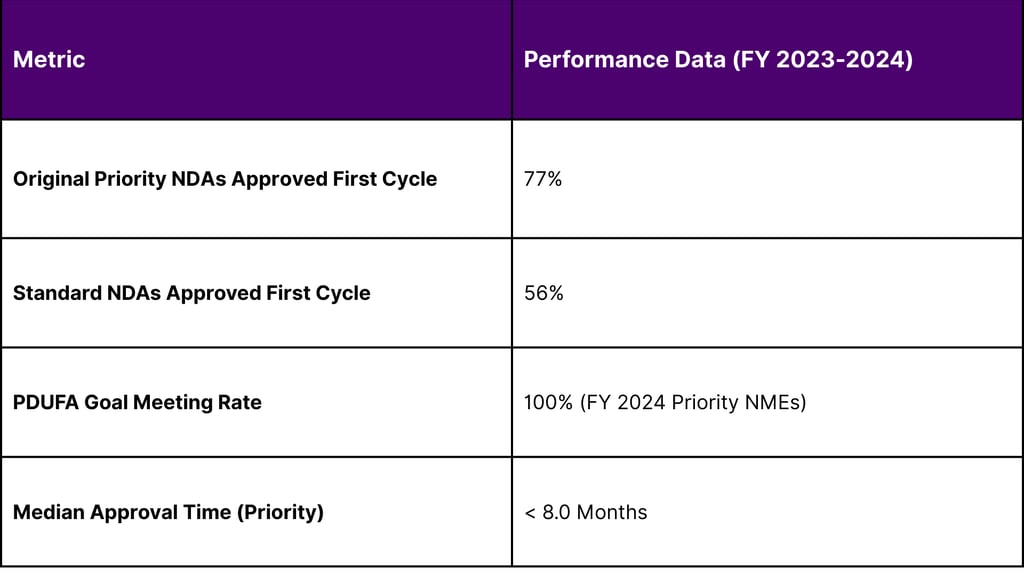
As of mid-March 2026, the rare disease market is increasingly dominated by ASOs due to their high target specificity and rapid development cycle. There are currently 14 FDA-approved ASO drugs, with over 50 candidates in Phase I-III trials and more than 100 in early-stage pipelines.
Recent Approval Milestones (2025-2026)
Tryngolza (Olezarsen): Approved Dec 19, 2024, to treat familial chylomicronemia syndrome by reducing triglycerides through APOC-III inhibition.
Dawnzera (Donidalorsen): Approved Aug 21, 2025, for the prevention of hereditary angioedema (HAE) attacks.
Wellcovorin (Leucovorin calcium): Approved March 10, 2026, as the first treatment for cerebral folate transport deficiency.
Zycubo (Copper histidinate): Approved Jan 12, 2026, for children with Menkes disease.
The orphan drug landscape continues to expand, with the FDA's Center for Drug Evaluation and Research (CDER) approving 46 novel drugs in 2025, a significant portion of which were for rare and ultra-rare conditions. Priority review vouchers and the Orphan Drug Act's tax incentives remain primary drivers for investment in the ASO space.
ASO Clinical Development Success Rates (Estimated 2024-2026)
The Integration of AI and Machine Learning
The acceleration of the ASO pipeline in 2026 is inseparable from the integration of artificial intelligence (AI). Platforms like eSkipFinder and ASOptimizer have transformed ASO design from an empirical screening process into a predictive science. These tools can predict the binding affinity and splicing efficiency of thousands of potential sequences in silico, reducing the reliance on extensive preclinical animal testing.
Furthermore, AI is being used to model ASO-protein interactions to predict potential liver and kidney toxicity. By analyzing the structural relationships of 2' modifications and their affinity for intracellular proteins, researchers can design "safer by design" oligonucleotides that avoid non-specific protein binding, a major cause of first-generation toxicity.
Future Directions: Multigene Targeting and Extracellular Vesicles
Looking beyond March 2026, the ASO field is exploring multigene RNA-based therapeutics. This strategy involves designing ASOs that can simultaneously silence multiple related genes, which may be essential for complex conditions like Alzheimer's Disease or cancer where compensatory pathways often lead to drug resistance.
Additionally, the use of extracellular vesicles (EVs) or exosomes for ASO delivery is gaining traction. A current clinical trial (NCT07382453) is evaluating a hydrogel-based delivery system for MSC-derived exosomes loaded with ASOs, aiming to provide a non-invasive, targeted approach for chronic diseases. These innovations, combined with the FDA's supportive regulatory framework, ensure that the ASO 2.0 revolution will continue to redefine the standard of care for rare genetic disorders for years to come.
Conclusions
The ASO 2.0 Revolution has fundamentally altered the therapeutic trajectory for patients with rare genetic disorders. Through the sophisticated application of third-generation chemistries like LNA and cEt, the development of targeted ligands such as GalNAc, and the implementation of AI-driven design, ASOs have overcome their historical limitations to become a dominant force in the market. The FDA's February 2026 Plausible Mechanism Framework marks the beginning of a new era where individualized medicine is no longer an exception but a scalable reality. As the field moves toward extrahepatic delivery and multigene targeting, the potential for ASOs to address the thousands of currently "untreatable" rare diseases remains one of the most promising frontiers in modern medicine.
Frequently Asked Questions (FAQ)
Q: What exactly is an "ASO 2.0" drug?
A: ASO 2.0 refers to the second and third generation of antisense oligonucleotides that use advanced sugar modifications like 2'-MOE and 2'-OMe or bridged structures like LNA. These modifications make the drugs more stable, more potent, and less toxic than the original first-generation phosphorothioate drugs.
Q: Why do some ASO treatments affect Vitamin A levels?
A: Some ASOs, like Wainua (Eplontersen), are designed to reduce the production of a protein called transthyretin (TTR). Because TTR is the carrier for Vitamin A in the blood, reducing TTR naturally lowers Vitamin A levels, which is why patients must take daily supplements.
Q: Is the new FDA framework only for ASOs?
A: While the February 2026 draft guidance specifically highlights RNA-based therapies like ASOs and genome editing, the "Plausible Mechanism Framework" is designed to be flexible and may apply to other tailored therapeutics that target the root cause of a disease.
Q: How are ASOs delivered to the brain?
A: Most ASOs for the brain are delivered via intrathecal injection a procedure where the drug is injected into the spinal canal. However, new research into non-viral vectors and "Mini-nucleosomes" aims to provide alternative, potentially less invasive delivery methods.
Q: What is the current success rate for ASO approvals?
A: According to recent FDA performance data, approximately 77% of priority applications for new molecular entities (which include many ASOs) were approved in their first review cycle in FY 2023.
References
1. Evolution of Antisense Oligonucleotides: Navigating Nucleic Acid ...
2. Antisense Oligonucleotides: An Emerging Area in Drug Discovery and Development - PMC
3. FDA Launches Framework for Accelerating Development of ...
4. Antisense oligonucleotides: a novel Frontier in pharmacological strategy - PMC
5. Drug Discovery Perspectives of Antisense Oligonucleotides - PMC
6. Splice-Modulating Antisense Oligonucleotides as Therapeutics for Inherited Metabolic Diseases - PMC
7. Pharmacokinetics and Proceedings in Clinical Application of Nucleic Acid Therapeutics
8. The chemical evolution of oligonucleotide therapies of clinical utility - PMC - NIH
9. Wainua - accessdata.fda.gov
10. Tofersen - StatPearls - NCBI Bookshelf - NIH
11. Artificial Intelligence-Driven Design of Antisense Oligonucleotides for Precision Medicine in Neuromuscular Disorders - PMC
12. Determining off-target effects of splice-switching antisense oligonucleotides using short read RNAseq in neuronally differentiated human induced pluripotent stem cells - PMC
13. CENTER FOR DRUG EVALUATION AND RESEARCH - accessdata.fda.gov
14. Nephrotoxicity of marketed antisense oligonucleotide drugs - PMC - NIH
15. Application of antisense oligonucleotide drugs in amyotrophic lateral sclerosis and Huntington's disease - PMC
16. Harnessing BET-Bromodomain Assisted Nuclear Import for Targeted Subcellular Localization and Enhanced Efficacy of Antisense Oligonucleotides - PMC
17. APPLICATION NUMBER: - 217388Orig1s000 OTHER REVIEW(S) - accessdata.fda.gov
18. Novel Drug Approvals for 2024 | FDA
19. GIVE Teaming Profiles - ARPA-H
20. Differential uptake, kinetics and mechanisms of intracellular trafficking of next-generation antisense oligonucleotides across human cancer cell lines - PMC
21. A therapeutic antisense oligonucleotide encompassing 2′-O-methoxyethyl modification triggers unique perturbation of the transcriptome - PMC
22. Tofersen for SOD1 ALS - PMC
23. Rare Disease Drug Approvals | FDA
24. 203568Orig1s000 - accessdata.fda.gov
25. RISE Workshop 2 - Individualized Therapies on the RISE - FDA
26. Study Details | NCT07363603 | Tianasen (ASO-GNAO1) for GNAO1-Encephalopathy With Epilepsy and Movement Disorders. | ClinicalTrials.gov
27. Novel Drug Approvals for 2025 | FDA
28. Guidance Documents for Rare Disease Drug Development | FDA
29. FY 2024 PDUFA Performance Report - Food and Drug Administration
30. Prescription Drug User Fee Act - FDA
31. Design, validation, and functional impact of oligonucleotides for multigene silencing in Alzheimer's disease - PMC
32. Study Details | NCT07382453 | Effect of PAE Hydrogel Loaded With tRF-ASO-Exo on Patients With Diabetic Ocular Surface Diseases | ClinicalTrials.gov

