The New Global Trial Map


The year 2026 represents a definitive inflection point in the global clinical research landscape. For decades, the biopharmaceutical industry operated within a transatlantic axis, with the United States and Western Europe conducting the vast majority of late-stage clinical trials. However, a convergence of geopolitical stability, sovereign wealth allocation, and regulatory maturation has firmly established the Gulf Cooperation Council (GCC) region specifically Saudi Arabia, the United Arab Emirates (UAE), and Qatar as a primary tier for clinical expansion. This report provides an exhaustive analysis of this paradigm shift, drawing exclusively from government data and regulatory announcements available as of February 2026.
The data reveals a market that has transcended its "emerging" status. In Saudi Arabia, the Saudi Food and Drug Authority (SFDA) reported an 83% surge in clinical trial applications for advanced therapies in 2025 alone. The UAE has sequenced over 700,000 genomes through the Emirati Genome Program, creating one of the world's most valuable datasets for rare disease target validation. Qatar, underpinned by a 2026 health budget of QAR 25.4 billion, has operationalized a National Reliance Agreement that dramatically reduces regulatory friction for multi-site oncology studies.
This report argues that the shift to the Middle East is driven by three "Unreported Accelerators":
The "Human Knockout" Advantage: High rates of consanguinity in the region provide a natural "human knockout" model, essential for validating gene targets in rare diseases a resource unavailable in the genetically heterogeneous populations of the West.
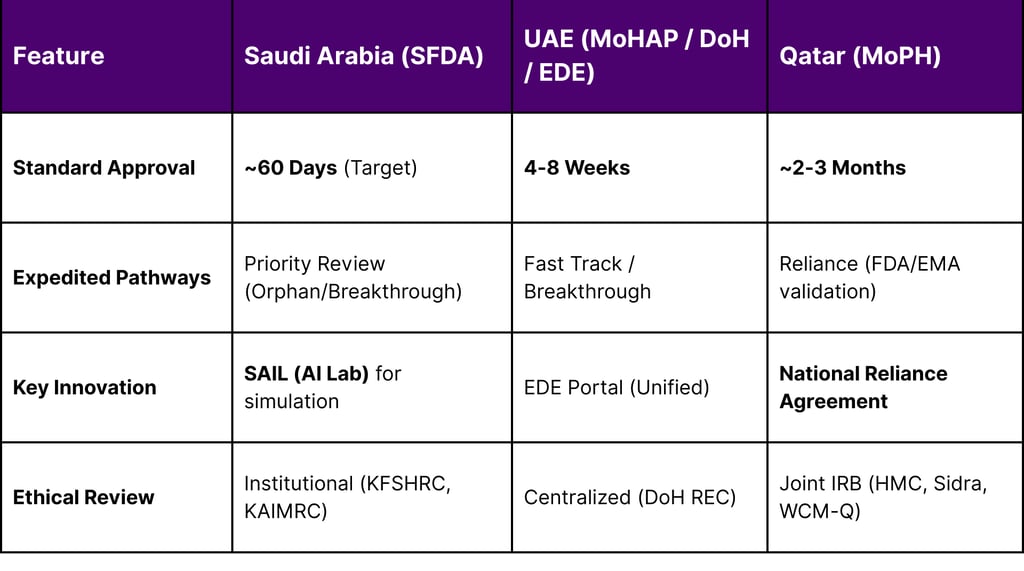
Regulatory Velocity: The harmonization of local regulatory timelines with FDA and EMA standards, utilizing reliance models and AI-driven review processes like the SFDA’s SAIL initiative.
Sovereign De-Risking: Direct government funding of the clinical infrastructure, such as Abu Dhabi’s 2026 Healthcare Research and Innovation Fund, which effectively subsidizes the high operational costs of complex oncology trials.
For biopharmaceutical sponsors, particularly in the high-stakes fields of Oncology and Rare Diseases, the Middle East in 2026 offers a solution to the twin crises of patient recruitment saturation and rising development costs.
Introduction: The 2026 Clinical Research Paradigm
The Global Saturation Crisis
By the mid-2020s, the traditional clinical trial hubs of North America and Western Europe began facing acute saturation. The proliferation of competing trials in oncology, specifically in PD-1/L1 inhibitors and CAR-T therapies, led to a scarcity of treatment-naïve patients. Recruitment rates slowed, and trial costs ballooned. Sponsors were forced to look beyond the "Tier 1" markets.
Simultaneously, the US Food and Drug Administration (FDA) issued stringent guidance on diversity in clinical trials, mandating that pivotal study data reflect the racial and ethnic diversity of the patient population intended to use the drug. This regulatory push compelled sponsors to seek diverse genomic backgrounds, making the homogenous populations of Eastern Europe less attractive and turning attention toward the ethnically diverse, treatment-naïve populations of the Middle East.
The Middle East Inflection Point
In 2026, the Middle East is no longer a passive recipient of medical innovation but an active co-developer. The governments of Saudi Arabia, UAE, and Qatar have pivoted their economic diversification strategies Saudi Vision 2030, We the UAE 2031, and Qatar National Vision 2030 toward the life sciences.
This is not merely about building hospitals; it is about constructing a "Research Economy." The region has moved from importing drugs to importing the science of drug development. This is evident in the strategic investments into Good Manufacturing Practice (GMP) facilities for cell therapy in Qatar and the integration of Artificial Intelligence (AI) into regulatory reviews in Saudi Arabia.
The result is a "New Global Trial Map" where Riyadh, Abu Dhabi, and Doha act as essential nodes in the global development network for breakthrough therapies.
Kingdom of Saudi Arabia: The Strategic Anchor
Saudi Arabia, the largest economy in the region, has undergone the most radical transformation. Under the Health Sector Transformation Program (HSTP), a pillar of Vision 2030, the Kingdom has restructured its entire healthcare apparatus to foster research and innovation.
The Scale of Growth: 2026 Statistics
The metrics for 2026 paint a picture of explosive growth. According to the SFDA's latest reports:
Clinical Trial Volume: There was an 83% year-on-year growth in clinical trial applications for advanced therapies and biotechnology products in 2025. This specific focus on "advanced therapies" (cell and gene therapies) indicates a qualitative shift from simple small-molecule trials to complex biologics.
Early Phase Expansion: Phase I and II trials, historically rare in the region due to safety liability concerns, saw a 39% increase. This suggests that global sponsors now trust the Kingdom’s safety monitoring and pharmacovigilance infrastructure enough to conduct "First-in-Human" studies.
Regulatory Evolution: SFDA Reforms
The Saudi Food and Drug Authority (SFDA) has evolved into a tier-one regulator. In 2026, the Authority implemented several key reforms designed to attract global sponsors.
The Orphan Drug Program
Recognizing the burden of rare genetic diseases, the SFDA operationalized its Orphan Drug Program. In January 2026, the Authority granted "Orphan Drug Designation" to several novel compounds, including Jascayd (nerandomilast) for Idiopathic Pulmonary Fibrosis (IPF) and Sephience (sepiapterin) for hyperphenylalaninaemia.
Mechanism: This designation provides sponsors with fee waivers, scientific advice, and, crucially, a priority review pathway. It signals a regulatory environment that actively incentivizes rare disease research.
Implication: For sponsors, this reduces the "regulatory risk" premium previously associated with the region.
AI-Driven Regulation (SAIL)
In a move to modernize its review capacity, the SFDA launched the SFDA Artificial Intelligence Laboratory (SAIL). As of January 2026, the SFDA CEO confirmed that AI is being used to simulate clinical trials and optimize data review processes.
Digital Twins: The Authority is utilizing "digital twin" technology to model regulatory outcomes, enhancing the predictability of approval timelines.
Efficiency: This reduces the administrative backlog, allowing the SFDA to maintain a competitive approval timeline of approximately 60 working days for standard trials, and significantly less for priority reviews.
Infrastructure for Advanced Therapies
The physical infrastructure in Saudi Arabia has expanded to meet the demands of complex trials.
CAR-T Capabilities: In August 2025, the SFDA approved the first Phase I clinical trial for a locally developed CAR-T cell therapy for Acute Lymphoblastic Leukemia (ALL) at King Faisal Specialist Hospital and Research Centre (KFSHRC). This trial is a proof-of-concept for the entire ecosystem demonstrating that Saudi hospitals can handle the apheresis, cryopreservation, and re-infusion logistics required for cell therapy.
Manufacturing Growth: The SFDA reported an 18% growth in licensed pharmaceutical factories in 2025. This localization of manufacturing capability is critical for the supply chain of investigational medicinal products (IMPs), reducing the reliance on costly and risky international shipments.
The 2026 Budgetary Landscape
The Saudi government’s commitment is financially codified. The 2026 budget continues to prioritize the Health Sector Transformation Program, with targets to increase healthcare service coverage to 88% and digitize 100% of medical records.
Unified Digital Records: The push for 100% digital medical record coverage 14 creates a centralized patient database. For clinical trial feasibility, this allows sponsors to query a national dataset to identify eligible patients in minutes rather than months.
United Arab Emirates: The Innovation Hub
The UAE has positioned itself as the "Silicon Valley" of healthcare in the region, focusing on high-tech, data-driven research initiatives.
The Emirati Genome Program (EGP)
The Emirati Genome Program is arguably the most significant research asset in the Middle East. By January 2025, the program had sequenced the genomes of 700,000 citizens, with a goal to cover the entire population.
Scientific Value: The EGP identified over 421 million single-nucleotide variants (SNVs), many of which are unique to the Arab population.
Clinical Application: This database allows for the identification of "founder mutations" responsible for rare diseases. Sponsors can use this data to validate drug targets or to find patients with specific genetic profiles for "basket trials" in oncology.
Admixture: The discovery that the Emirati population is an admixture of African, Asian, and European ancestries makes it an ideal proxy for global diversity, satisfying FDA diversity mandates.
Abu Dhabi Department of Health (DoH) Initiatives
Abu Dhabi acts as the primary engine for the UAE’s life sciences strategy.
Research Funding: The 2026 Healthcare Research and Innovation Fund was launched to support translational research. In the 2024-2025 cycle, grants were awarded to projects focused on immunologic disorders and cancer prevention. This funding mechanism de-risks early-stage research for academic and private partners.
HTA Guidelines: The introduction of Health Technology Assessment (HTA) Guidelines in mid-2025 provided a transparent framework for reimbursement. This is critical for sponsors of high-cost gene therapies, as it outlines exactly how the government evaluates "value," enabling better commercial planning alongside clinical development.
Institutional Centers of Excellence
Sheikh Shakhbout Medical City (SSMC): As of 2026, SSMC is a premier site for complex trials. It was selected as the only site in the UAE for a multinational CAR-T trial and one of only two centers in the Middle East for a gene therapy trial. The hospital also runs AI-driven studies in cardiology and endocrinology, leveraging its partnership with the Mayo Clinic.
Cleveland Clinic Abu Dhabi: This facility continues to lead in oncology research, conducting genome-based studies on breast health and obesity specifically tailored to the Emirati population.
Regulatory Harmonization: The Emirates Drug Establishment
The creation of the Emirates Drug Establishment (EDE) has streamlined the regulatory landscape. By consolidating the regulatory functions previously held by the Ministry of Health and Prevention (MoHAP), the EDE provides a "single window" for clinical trial applications. This reduces the bureaucratic friction between federal and emirate-level approvals (e.g., DoH vs. DHA), accelerating study start-up times.
Qatar: The Specialist in Quality and Precision
Qatar’s approach is characterized by focused, high-investment specialization. Rather than aiming for volume, Qatar targets high-complexity "niche" trials that leverage its world-class academic medical centers.
Qatar Cancer Plan 2023-2026
The Qatar Cancer Plan 2023-2026, titled "Excellence for All," serves as the roadmap for oncology research.
Precision Medicine: The plan explicitly mandates the development of an operating model for precision medicine services (Deliverable 3.3.C). This ensures that every cancer patient is potentially a research participant, with their tumor profiled for genetic mutations.
Comprehensive Care: The plan includes the development of a new Comprehensive Cancer Center, integrating research directly into clinical care pathways.
Governance: The establishment of a National Cancer Program team within the Ministry of Public Health (MoPH) ensures that research priorities are aligned with national health needs.
Fiscal Power: The 2026 Budget
Qatar’s commitment to health research is backed by substantial sovereign wealth. The 2026 general budget allocated QAR 25.4 billion to the health sector, a 15.4% increase from 2025.
Allocation: This funding supports the expansion of the Hamad Medical Corporation (HMC) and the research initiatives of the Qatar Foundation.
Grant Funding: The Qatar Research, Development, and Innovation (QRDI) Council launched the "Road to Biopharma" call for 2026. This grant provides QAR 300,000 per project to strengthen the regulatory and infrastructure ecosystem for biopharmaceuticals.
Research Infrastructure: Sidra and HMC
Sidra Medicine: Sidra is a global leader in pediatric rare disease research. Its GMP facility is licensed to manufacture individualized therapeutic products, including gene therapies. In 2026, Sidra was a recipient of the QRDI Technology Development Grant, highlighting its role in commercializing medical innovation.
National Reliance Agreement: To tackle the bottleneck of multi-site approvals, HMC, Sidra Medicine, and Weill Cornell Medicine-Qatar operationalized a National Reliance Agreement. This allows a trial approved by one institution’s IRB to be accepted by the others, significantly reducing start-up timelines for multi-center studies.
The Genomic & Demographic Advantage
Beyond infrastructure and regulation, the Middle East offers an intrinsic "biological asset" that is driving its rise as a research hub: its people.
The "Human Knockout" Phenomenon
Consanguinity (marriage between close relatives) remains culturally common in parts of the GCC. Scientifically, this increases the probability of autosomal recessive disorders, where a child inherits two copies of a mutated gene.
Research Value: This creates "human knockouts" individuals who completely lack the function of a specific gene. Studying these individuals allows researchers to understand the physiological effect of inhibiting that gene, which is exactly what many drugs aim to do.
Validation: The Emirati Genome Program and Qatar Genome Program are actively mining their datasets for these loss-of-function variants. For a pharmaceutical company developing a drug to inhibit Protein X, finding a human who naturally lacks Protein X is the ultimate validation of safety and efficacy.
Diversity and the FDA Mandate
With the US FDA requiring diversity action plans for clinical trials, the Middle East offers a solution.
Admixture: The population of the Arabian Peninsula is not genetically monolithic. As shown by the EGP, it contains distinct ancestry components ("Arabian I" and "Arabian II") that are underrepresented in global databases like the 1000 Genomes Project.
Expatriate Diversity: The workforce in the GCC includes millions of South Asians, Southeast Asians, and Africans. This allows sponsors to recruit a globally diverse cohort within a single geographic location.
Disease Prevalence as a Driver
The region suffers from a unique epidemiological profile.
Rare Diseases: Due to genetic factors, the prevalence of rare metabolic and genetic disorders is significantly higher than in the West.
Oncology: There is a rising incidence of early-onset cancers (breast, colorectal) in the region.
Lifestyle Diseases: High rates of diabetes and obesity make the region a prime location for metabolic disease trials, which often overlap with oncology (e.g., metabolic drivers of cancer).
Comparative Regulatory & Operational Analysis
Sponsors considering the Middle East in 2026 must understand the operational nuances between the three major hubs.
Regulatory Timelines and Pathways (2026 Status)
Data Privacy and Sovereignty
Data localization is a critical consideration in 2026.
UAE Health Data Law: Federal Law No. 2 restricts the transfer of health data outside the UAE. However, exceptions for scientific research are available and clearly defined. Sponsors must plan for local data storage or secure federated access.
Saudi Data Sovereignty: The National Data Management Office (NDMO) enforces strict data residency. The SFDA’s investment in local digital infrastructure supports this, requiring sponsors to use local cloud providers for trial data management.
Logistics and Supply Chain
Saudi Arabia: The 18% growth in licensed factories and partnerships with the Saudi Logistics Academy have strengthened the cold chain, essential for transporting temperature-sensitive biologics like CAR-T cells.
UAE: As a global aviation hub, the UAE offers the most established logistics network. The presence of major courier hubs (DHL, FedEx, UPS) in Dubai and Abu Dhabi ensures 24-hour connectivity to central labs in Europe or the US, although local central lab capacity is growing.
Therapeutic Deep Dives
Oncology: The Primary Driver
Oncology trials dominate the regional landscape, accounting for the largest share of interventional studies.
Immuno-Oncology: The region is actively participating in global Phase III trials for checkpoint inhibitors. The diverse genetic background of patients helps identify responders vs. non-responders.
Cell Therapy: The KFSHRC CAR-T trial and SSMC’s multinational CAR-T study demonstrate that the region has crossed the "capability gap." They can now perform the complex bedside procedures required for these therapies.
Preventive Oncology: Abu Dhabi’s 2026 funding for "innovation in cancer prevention" opens the door for trials on early detection biomarkers and liquid biopsies.
Rare Diseases: The Niche Opportunity
The "Orphan Drug" market is the fastest-growing segment in the region.
Gene Therapy: With Sidra Medicine’s GMP facility and SSMC’s gene therapy trials, the region is becoming a hub for "n-of-1" trials and ultra-rare disease studies.
Metabolic Disorders: The high prevalence of PKU and other metabolic conditions has led to specific drug designations like Sephience, indicating a robust pipeline of metabolic trials.
Challenges and Future Outlook
While the trajectory is positive, challenges remain that sponsors must mitigate.
Workforce Capacity
The rapid expansion of trials has created a demand for qualified Clinical Research Associates (CRAs) and Principal Investigators (PIs).
Mitigation: The Saudi Health Sector Transformation Program involves massive training initiatives. Qatar’s QRDI grants also mandate capacity building. Sponsors are increasingly partnering with Contract Research Organizations (CROs) that have established local training academies.
Patient Recruitment Awareness
While the patient pool exists, awareness of clinical trials can be low.
Mitigation: The UAE’s "We the UAE 2031" and Saudi’s "Quality of Life" programs include public health education components. Digital recruitment campaigns, leveraging the region’s extremely high smartphone penetration, are proving effective.
Long-Term Follow-Up
For gene therapies, patients must be followed for 15+ years.
Mitigation: The digitization of medical records (100% target in KSA) and platforms like "Emirates Health" provide the digital backbone necessary for long-term real-world evidence (RWE) generation, ensuring patients are not "lost to follow-up."
Conclusion: The Strategic Imperative for 2026
The data from early 2026 confirms that the Middle East has successfully transitioned from an emerging market to a priority region for global clinical research. The aggressive regulatory reforms by the SFDA, the massive genomic investments by the UAE, and the specialized infrastructure funding in Qatar have created an ecosystem that rivals traditional Western hubs in quality, speed, and capability.
For biopharmaceutical sponsors, the Middle East offers a strategic solution to global development bottlenecks. It provides access to:
Untapped, genetically diverse patient populations.
State-of-the-art infrastructure subsidized by sovereign wealth.
Regulatory pathways designed for speed and flexibility.
In the competitive race to develop the next generation of oncology and rare disease therapies, inclusion of the Middle East in the global trial footprint is no longer optional it is a competitive necessity.
Frequently Asked Questions (FAQ)
Q1: What are the regulatory timelines for clinical trial approval in Saudi Arabia in 2026?
A: The SFDA has significantly streamlined its processes. For standard trials, the target is approximately 60 days. However, for Orphan Drugs or Breakthrough Therapies, expedited pathways exist that can shorten this timeline. The introduction of AI-assisted review via the SAIL lab aims to further reduce administrative bottlenecks.
Q2: Can patient samples be exported from the UAE for central lab analysis?
A: Generally, UAE Health Data Law restricts the export of biological samples and data. However, exceptions are granted for scientific research and clinical trials. Sponsors must obtain specific approval from the relevant health authority (DoH or DHA) and typically need to justify why the analysis cannot be performed locally. The growth of local central labs is reducing the need for export.
Q3: Is it mandatory to include local nationals in clinical trials in Qatar?
A: While not explicitly mandatory for all phases, the Qatar Cancer Plan and MoPH guidelines strongly encourage the inclusion of the local population to ensure the genetic relevance of the data. For genomic studies, the Qatar Genome Program focuses specifically on the Qatari population to build a reference map.
Q4: How does the Emirati Genome Program benefit foreign pharmaceutical sponsors?
A: The EGP offers a massive reference database that helps in validating drug targets, particularly for rare diseases. By understanding the specific genetic variants present in the population, sponsors can design more effective inclusion/exclusion criteria and identify patients with specific biomarkers that might be rare elsewhere.
Q5: Are there incentives for conducting rare disease trials in the region?
A: Yes. Saudi Arabia’s SFDA offers an Orphan Drug Designation which includes incentives like priority review and extended market exclusivity periods post-approval. Qatar’s QRDI Council offers grants (e.g., Road to Biopharma) to support the infrastructure and regulatory costs for such projects.
Q6: How does the "National Reliance Agreement" in Qatar work?
A: It is a collaborative framework between Hamad Medical Corporation (HMC), Sidra Medicine, and Weill Cornell Medicine-Qatar. It allows a clinical trial protocol approved by the IRB of one institution to be accepted by the others without a full de novo review, streamlining the approval process for multi-site studies within the country.
References
SFDA: 83% Growth in Clinical Research for Biotechnology Products .
The UAE Genome Program: Unique Genetic Insights from 43,608 .
His Highness the Amir approves 2026 general budget.
REPORT - Hamad Medical Corporation.
News | The official website of the Saudi Food and Drug Authority.
Ma'an Invite Researchers and Innovators to Apply for the 2026 Healthcare Research and Innovation Fund - News | Department of Health - Abu Dhabi.
Diversity Action Plans to Improve Enrollment of Participants from Underrepresented Populations in Clinical Studies | FDA, accessed on February 4, 2026.
Medical Research Archives - Sidra Medicine.
Drugs | The official website of the Saudi Food and Drug Authority.
SFDA - Saudi Food And Drug Authority - Medrais.
INCENTIVES OF ORPHAN DRUG DESIGNATION.
SFDA Timelines - PharmaKnowl.
SFDA Approves First-of-its-Kind Clinical Trial for a Gene Therapy Developed in Saudi Arabia to Treat Acute Lymphoblastic Leukemia (ALL).
Saudi Vision 2030 - Health Sector Transformation Program.
In collaboration with Authority of Social Contribution – Ma'an, Department of Health – Abu Dhabi opens applications for 2026 Healthcare Research and Innovation Fund.
Abu Dhabi Health Technology Assessment Guidelines - Ronin Legal.
Research Highlights & News - Sheikh Shakhbout Medical City.
Research News – Sheikh Shakhbout Medical City Abu Dhabi.
Cleveland Clinic Abu Dhabi named top research hospital in UAE for second year running.
News | Ministry of Health and Prevention - UAE.
Qatar Cancer Plan 2023-2026 - ICCP Portal.
QRDI/QNRF Funding Opportunities - Georgetown University in Qatar.
Web Summit Qatar 2026: QRDI Council Announces Technology.
Qatar Genome Program – GA4GH.
UAE Medical Device Registration | MoHAP Guide 2025 - Kayrouz & Associates.
United Arab Emirates - Healthcare and Life Science - International Trade Administration.
Transforming healthcare: Saudi Arabia's vision 2030 healthcare model - PMC.
Home | Ministry of Health and Prevention - UAE - وزارة الصحة ووقاية المجتمع.

