The Radiopharma Acceleration


The radiopharmaceutical sector is experiencing an unprecedented transformation, with targeted alpha therapies (TATs) positioned at the forefront of precision oncology. As we enter 2026, the convergence of regulatory clarity, clinical validation, and manufacturing scale-up signals a definitive breakout year for this therapeutic modality. This analysis examines the regulatory landscape, clinical trial activity, and market dynamics driving radiopharmaceutical acceleration.
The Regulatory Renaissance: FDA Sets New Standards
Draft Guidance Reshapes Development Pathways
In August 2025, the FDA issued draft guidance titled "Oncology Therapeutic Radiopharmaceuticals: Dosage Optimization During Clinical Development," marking a watershed moment for the sector. This guidance, which closed for public comment on October 20, 2025, provides unprecedented clarity on clinical development expectations for radiopharmaceutical therapies.
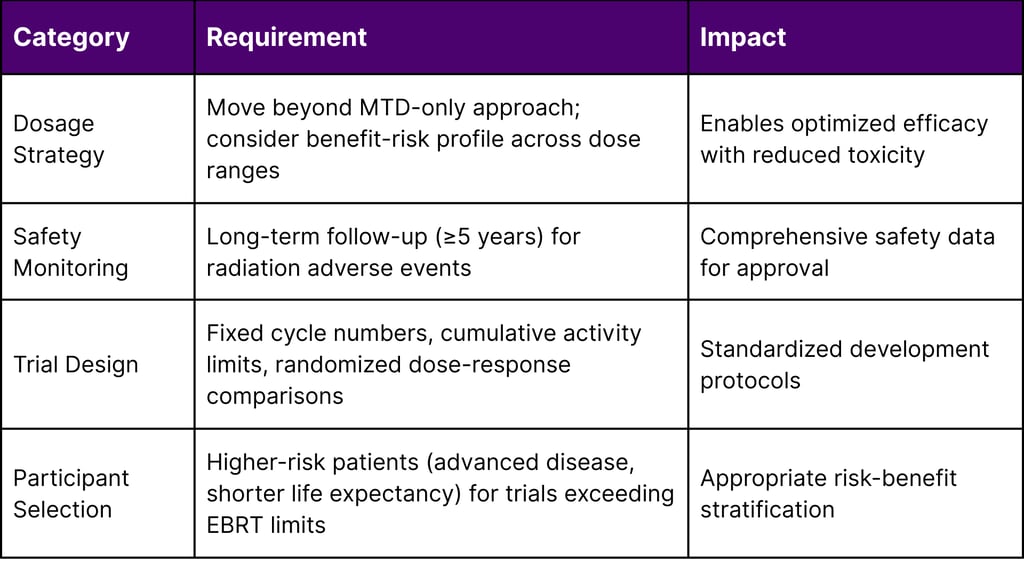
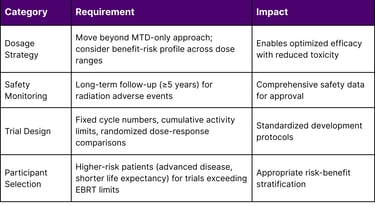
The guidance addresses critical developmental challenges unique to radiopharmaceuticals, including:
Dosage optimization strategies that extend beyond traditional maximum tolerated dose (MTD) approaches
Safety monitoring protocols for delayed, cumulative, and irreversible toxicity
Dosimetry requirements for clinical trials
Participant selection criteria for trials exceeding external beam radiation therapy (EBRT) organ tolerances
Table 1: Key FDA Guidance Provisions for Radiopharmaceutical Development
This regulatory framework complements the FDA's August 2024 guidance on "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," creating a comprehensive roadmap for radiopharmaceutical development.
Breakthrough Therapy Designations Signal Regulatory Confidence
The FDA's grant of Breakthrough Therapy Designation to AlphaMedix (lead-212 DOTAMTATE) in February 2024 for peptide receptor radionuclide therapy (PRRT)-naïve patients with gastroenteropancreatic neuroendocrine tumors represents a pivotal regulatory endorsement of targeted alpha therapy. This designation, reserved for therapies demonstrating substantial improvement over existing treatments, accelerates development timelines and increases regulatory interaction.
Clinical Trial Landscape: Unprecedented Expansion
Phase 3 Trial Proliferation
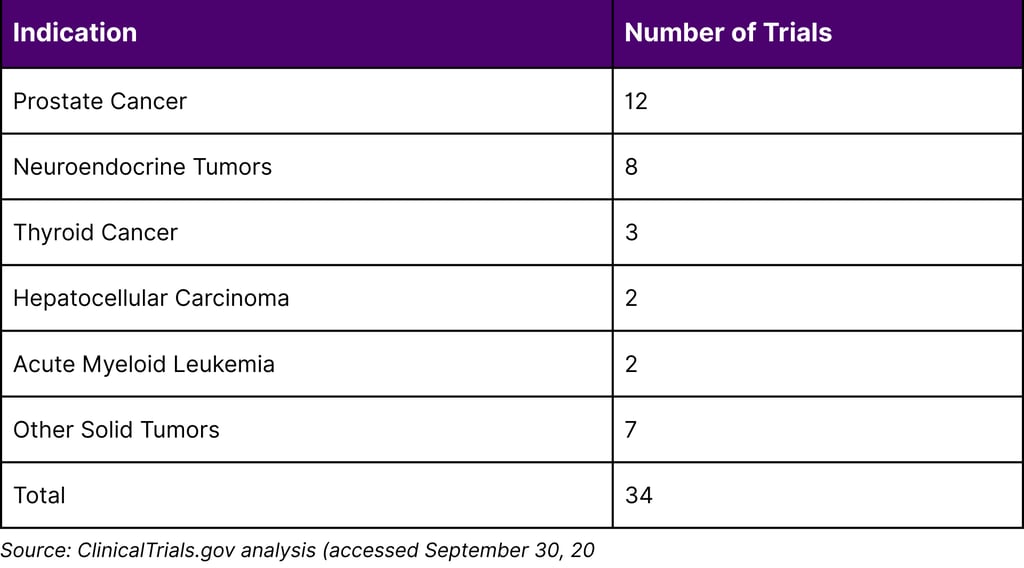
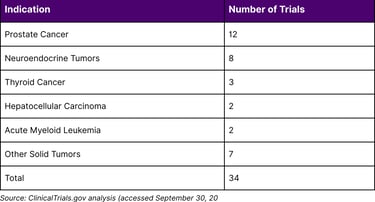
As of September 2024, 34 active Phase 3 radiopharmaceutical therapy trials are registered on ClinicalTrials.gov, representing a dramatic expansion from previous years. These trials span diverse oncological indications:
Figure 1: Active Phase 3 Radiopharmaceutical Trials by Indication (2024)
Lutetium-177: The Current Gold Standard
Lutetium-177, with over 200 ongoing clinical studies, has emerged as the dominant radioisotope in therapeutic applications. Its favorable nuclear properties including a 6.7-day half-life and appropriate beta-emission energy have positioned it as the benchmark for radiopharmaceutical therapy development.
Table 2: FDA-Approved Lutetium-177 Radiopharmaceuticals
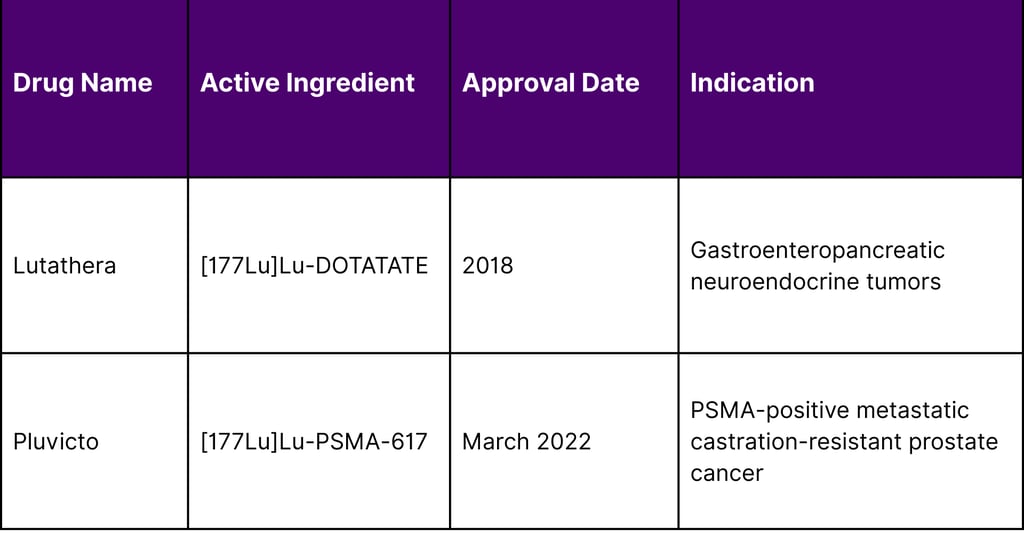
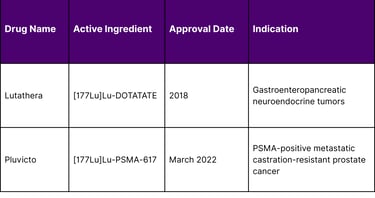
The March 2025 FDA approval expansion for Pluvicto, allowing treatment following androgen receptor pathway inhibitor therapy before chemotherapy, is projected to triple eligible patient populations from approximately 10,000 to 30,000 patients annually in the United States.
Targeted Alpha Therapy: The Next Frontier
Alpha Emitters Demonstrate Superior Cytotoxic Potential
Targeted alpha therapies utilize alpha-emitting radionuclides that deliver high linear energy transfer radiation over extremely short ranges (50-100 micrometers), equivalent to just a few cell diameters. This physical property enables precise tumor cell destruction while minimizing damage to adjacent healthy tissue.
Table 3: Leading Alpha-Emitting Radionuclides in Clinical Development
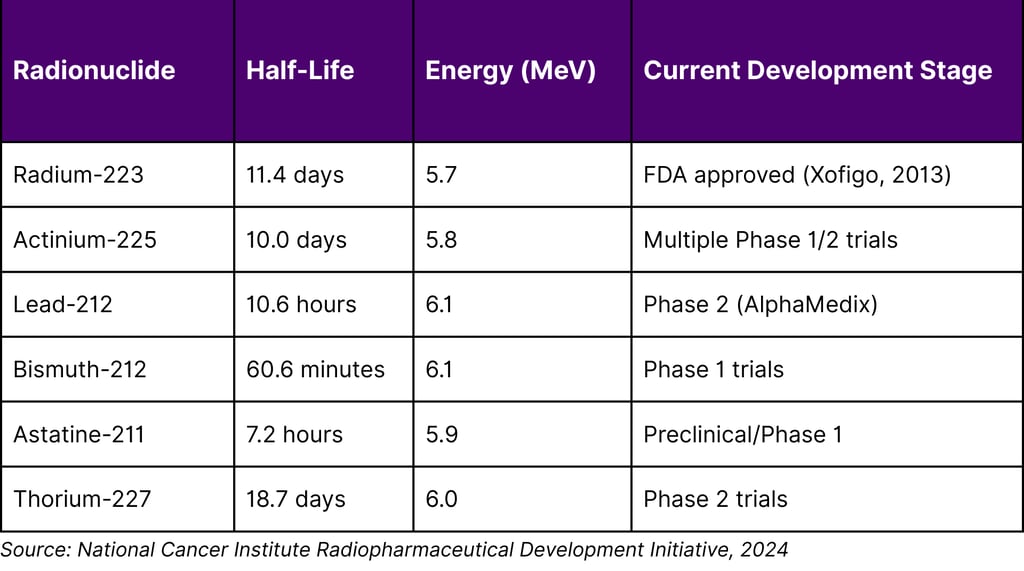
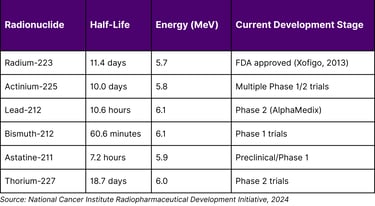
Landmark Alpha Therapy Clinical Results
AlphaMedix Phase 2 Study (ALPHAMEDIX-02)
In October 2025, Sanofi reported that AlphaMedix achieved all primary efficacy endpoints in the Phase 2 ALPHAMEDIX-02 study for patients with gastroenteropancreatic neuroendocrine tumors. Key findings included:
Clinically meaningful overall response rates in both PRRT-naïve and PRRT-exposed cohorts
Prolonged progression-free survival benefits
Manageable safety profile across both patient populations
Demonstrated activity in patients previously treated with lutetium-177 DOTATATE
These results form the basis for ongoing discussions with health authorities regarding potential regulatory submissions.
Xofigo: Pioneering Alpha Therapy Success
Radium-223 dichloride (Xofigo), approved by the FDA in 2013 for castration-resistant prostate cancer with bone metastases, validated the clinical potential of targeted alpha therapy. The PEACE-3 trial data presented at ESMO 2024 demonstrated that combining Xofigo with enzalutamide improved radiographic progression-free survival in untreated metastatic castration-resistant prostate cancer, supporting potential first-line treatment positioning.
Manufacturing and Supply Chain Evolution
Addressing the Alpha Emitter Supply Challenge
Historic supply constraints for alpha-emitting isotopes have been a primary development bottleneck. However, significant infrastructure investments are addressing this limitation:
Infrastructure Developments:
Orano Med's Production Expansion: Construction of the Indianapolis ATLab facility completed in mid-2024, enabling lead-212 production throughout North America
TerraPower Isotopes: U.S. production facilities positioned to meet increasing actinium-225 demand
SHINE Technologies: Domestic radioisotope production capability supporting clinical trial supply
International Atomic Energy Agency (IAEA): Focused initiatives on three priority alpha-emitting radionuclides to support global demand
CDMO Sector Acceleration
Contract Development and Manufacturing Organizations specializing in radiopharmaceuticals are experiencing rapid expansion. PharmaLogic's April 2025 acquisition of Agilera CDMO demonstrates industry consolidation aimed at achieving commercial-scale manufacturing capability.
Cancer Epidemiology Context
The Oncology Treatment Gap
According to the American Cancer Society's 2025 cancer statistics, an estimated 2.05 million new cancer cases will be diagnosed in the United States in 2025. Despite steady progress, with overall cancer death rates declining 1.5% annually from 2018 to 2022, significant unmet medical needs persist across multiple cancer types.
Table 4: U.S. Cancer Incidence and Mortality Rates (2021-2022)
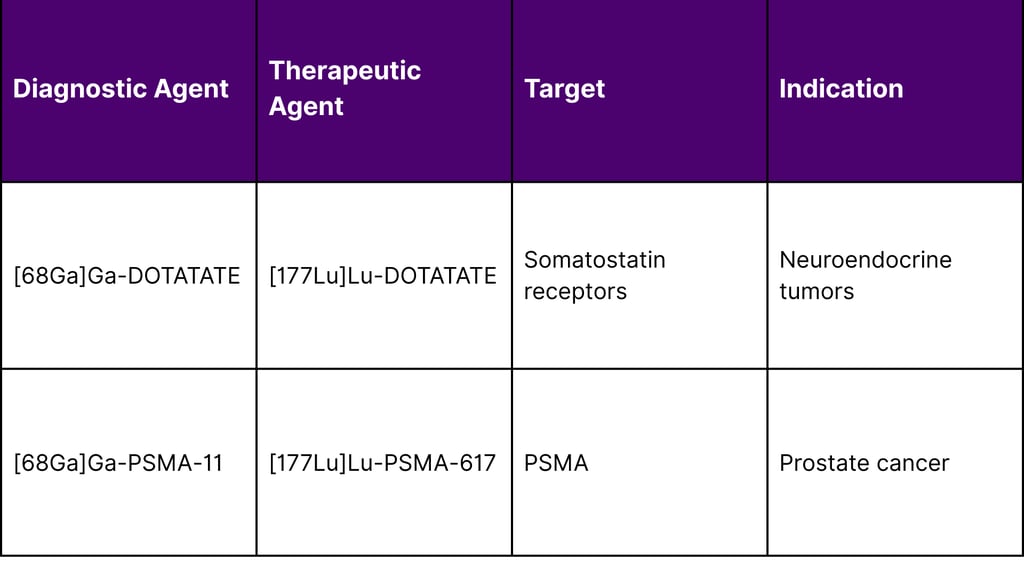
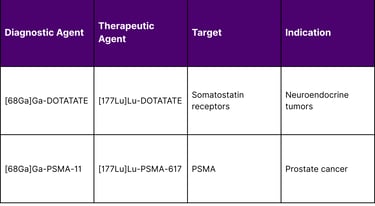
Patient Selection Optimization
Diagnostic radiopharmaceutical imaging enables identification of patients most likely to benefit from therapy based on target expression levels, tumor burden distribution, and predicted radiation dosimetry. This precision approach maximizes therapeutic benefit while minimizing exposure in non-responding patients.
2026 Catalysts: Why This Year Matters
Regulatory Finalization
The finalization of FDA guidance on radiopharmaceutical dosage optimization, expected in 2026, will provide definitive development pathways for sponsors. This regulatory clarity reduces development risk and accelerates clinical trial initiation.
Phase 3 Data Maturation
Multiple pivotal Phase 3 trials are scheduled to report results in 2026, including:
Alpha therapy studies in neuroendocrine tumors
PSMA-targeted therapy line extensions
Novel isotope-ligand combinations across multiple indications
Manufacturing Scale-Up
The operationalization of expanded production facilities for alpha-emitting isotopes in 2025-2026 will alleviate supply constraints that have historically limited clinical trial enrollment and commercial availability.
Combination Therapy Evidence
The National Cancer Institute's Radiopharmaceutical Development Initiative, launched in December 2024, focuses on early-phase combination studies testing radiopharmaceuticals with novel agents including immunotherapies. Initial data from these studies will emerge in 2026, potentially expanding treatment applications.
Challenges and Considerations
Long-Term Safety Monitoring
The FDA's emphasis on ≥5-year safety follow-up for radiation adverse events reflects appropriate caution regarding potential delayed toxicities. Comprehensive long-term data will be essential for broad therapeutic acceptance.
Health Care Delivery Infrastructure
Radiopharmaceutical administration requires specialized facilities, trained personnel, and radiation safety protocols. Expansion beyond major academic medical centers to community oncology settings will require infrastructure investment and workforce training.
Reimbursement Pathways
While Medicare coverage exists for FDA-approved radiopharmaceuticals, reimbursement adequacy and prior authorization requirements vary. Clear health economic evidence demonstrating value relative to existing treatments will facilitate broader access.
Strategic Implications
For Pharmaceutical Developers
The radiopharmaceutical opportunity requires:
Strategic partnerships with isotope suppliers and CDMOs
Theranostic development programs combining diagnostic and therapeutic agents
Clinical trial designs aligned with FDA guidance expectations
Long-term safety data collection infrastructure
For Healthcare Systems
Oncology programs should evaluate:
Radiopharmaceutical therapy capabilities and infrastructure needs
Multidisciplinary team development (nuclear medicine, radiation oncology, medical oncology)
Dosimetry and radiation safety protocols
Patient selection and referral pathways
For Investors and Strategic Acquirers
Due diligence priorities include:
Manufacturing capability and isotope supply security
Clinical development stage and regulatory pathway clarity
Target selection and competitive positioning
Intellectual property portfolio strength
Conclusion: A Transformative Moment
The radiopharmaceutical sector, and particularly targeted alpha therapy, stands at an inflection point. The convergence of regulatory clarity through FDA guidance, expanding clinical evidence from Phase 3 trials, manufacturing infrastructure maturation, and substantial industry investment positions 2026 as the breakout year for this therapeutic modality.
For patients facing difficult-to-treat cancers, radiopharmaceuticals offer precision-targeted therapy with improved benefit-risk profiles compared to traditional approaches. For the oncology ecosystem pharmaceutical companies, CDMOs, healthcare systems, and strategic investors radiopharmaceuticals represent a generational opportunity to advance cancer care.
As the field accelerates, stakeholders who establish positions now, aligned with regulatory expectations and clinical evidence standards, will be best positioned to capture value in this transformative market.
Frequently Asked Questions
Q1: What distinguishes targeted alpha therapy from other radiopharmaceuticals?
Alpha-emitting radionuclides deliver high linear energy transfer radiation over extremely short ranges (50-100 micrometers), enabling precise tumor cell destruction with minimal damage to surrounding healthy tissue. This differs from beta-emitters like lutetium-177, which have longer tissue penetration ranges.
Q2: How many radiopharmaceutical clinical trials are currently active?
As of September 2024, there are 34 active Phase 3 radiopharmaceutical therapy trials registered on ClinicalTrials.gov, with over 200 total studies involving lutetium-177 alone across all phases.
Q3: What is the FDA's position on radiopharmaceutical dosing that exceeds traditional radiation therapy limits?
The FDA's August 2025 draft guidance indicates that dosages exceeding external beam radiation therapy organ tolerances may be justified based on clinical data, provided trials include appropriate safeguards such as participant selection, trial design, safety monitoring, and dosimetry evaluation.
Q4: Which cancer types are primary targets for radiopharmaceutical development?
Prostate cancer and neuroendocrine tumors currently lead radiopharmaceutical development, with FDA-approved therapies in both indications. Expanding applications include thyroid cancer, hepatocellular carcinoma, acute myeloid leukemia, and various solid tumors expressing targetable biomarkers.
Q5: What is driving the recent surge in pharmaceutical industry investment in radiopharmaceuticals?
Key drivers include: (1) clinical validation through FDA approvals of Lutathera and Pluvicto, (2) regulatory clarity via new FDA guidance, (3) expanding eligible patient populations through earlier treatment line positioning, (4) theranostic precision medicine advantages, and (5) favorable competitive dynamics in high-unmet-need indications.
Q6: How do radiopharmaceuticals fit into the broader precision oncology landscape?
Radiopharmaceuticals exemplify precision medicine through molecular target-based patient selection (via companion diagnostic imaging), precise radiation delivery to cancer cells, and real-time treatment response monitoring. This approach aligns with the broader oncology trend toward biomarker-driven, personalized therapeutic strategies.
Q7: What are the main supply chain challenges for radiopharmaceuticals?
The primary challenge has been limited production capacity for alpha-emitting isotopes, which have historically been available only in small quantities from specialized facilities. However, significant infrastructure investments in 2024-2025, including new production facilities in North America and Europe, are addressing this bottleneck.
Q8: What role does the National Cancer Institute play in radiopharmaceutical development?
The NCI's Radiopharmaceutical Development Initiative, launched in December 2024, provides specialized infrastructure for clinical evaluation of novel theranostic radiopharmaceutical cancer therapies, with particular focus on early-phase combination studies with other novel agents.
References
American Cancer Society. (2025). Cancer statistics, 2025. CA: A Cancer Journal for Clinicians.
National Cancer Institute. (2025). Annual report to the nation on the status of cancer. National Institutes of Health.
U.S. Food and Drug Administration. (2024). Advancing health through innovation: New drug therapy approvals 2024. Center for Drug Evaluation and Research.
U.S. Food and Drug Administration. (2025). Novel drug approvals for 2025. Center for Drug Evaluation and Research.
U.S. Food and Drug Administration. (2025). Oncology therapeutic radiopharmaceuticals: Dosage optimization during clinical development [Draft guidance]. Center for Drug Evaluation and Research.
U.S. Food and Drug Administration. (2025, August 19). Oncology therapeutic radiopharmaceuticals: Dosage optimization during clinical development; Draft guidance for industry; Availability. Federal Register, 90(159), 40370-40371.
Lepareur, N. (2025). An overview of current phase 3 radiopharmaceutical therapy clinical trials. Frontiers in Medicine, 12, 1549676.
Zhang, S., Wang, X., Gao, X., et al. (2025). Radiopharmaceuticals and their applications in medicine. Signal Transduction and Targeted Therapy, 10(1), 1.
Sanofi. (2025, October 8). AlphaMedix™ (212Pb-DOTAMTATE) achieved all primary efficacy endpoints in phase 2 study [Press release].

